Rising living costs and pollution may have made you consider opting for solar at home or your business.
Thin-film solar panels are worth considering since they are cheaper than ordinary silicon solar panels.
This article will cover all aspects of thin-film solar panels.
Table of Contents
What Are Thin-Film Solar Panels?
To put it simply, thin-film solar panels are a type of PV panel belonging to the broader solar panel family.
But let’s dig a little deeper.
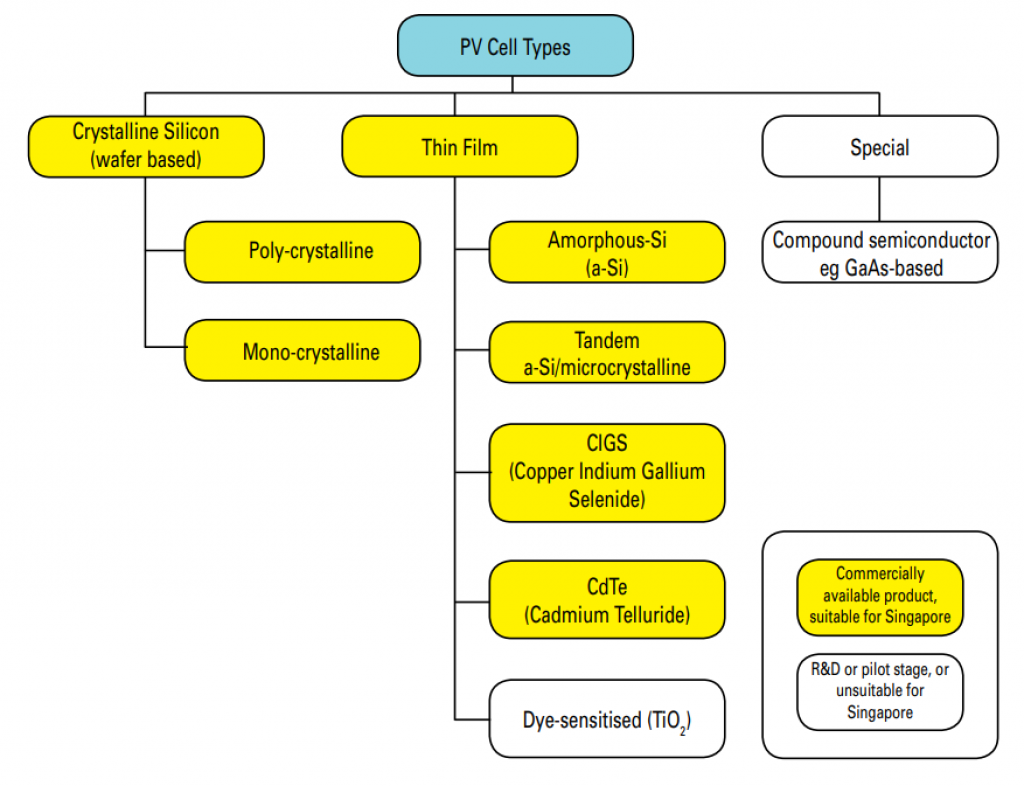
Source: Singapore Energy Market Authority
Thin-Film Cells Vs. Silicon Cells
Solar cells are the basic electricity-producing unit of a panel.
Most solar cells are crystalline silicon solar cells made from thin wafers. These wafers are typically 150 to 200 microns thick (i.e., 2/1,000ths of an inch).
You might see black or blue solar panels on the rooftops of houses in your suburb. They are either polycrystalline silicon or monocrystalline silicon solar panels. They are called “crystalline” because the silicon is organized into a crystal structure.
Mono-crystalline silicon cells are made from a single ingot of silicon. This ingot is very carefully cast to produce a single crystal structure.
Polycrystalline silicon is produced quicker and so is cheaper. Instead of one crystal, it contains many crystals, each tiny in size. For that reason, monocrystalline and polycrystalline silicon cells look different and differ in price.

Source: Climatebiz
In contrast, thin-film cells are made of ultra-thin layers of semiconductors. These layers are applied onto a glass or stainless steel substrate, only 0.3 to 2 microns thick (i.e., 2/100,00ths of an inch).
This is how “thin-film” cells got their name. They are 200 or 300 times thinner than silicon solar cells.
Thin-film solar cells cost less than silicon cells because making them requires less material.
How Do Thin-Film Solar Panels Work?
Semiconductors, Insulators, Conductors
Solar cells generate electricity when exposed to light. But why does this happen?
Firstly, we need to define conductivity, conductors, and insulators.
- Conductivity: electrical conductivity measures how easily electricity flows through a material.
- Conductors: copper is an excellent electrical conductor: its conductivity is very high. For this reason, we use it to make electrical cables.
- Insulators: plastic is generally a poor electrical conductor. As such, we use it as housing for household appliances. It’s an insulator.
Semiconductors are neither insulators nor conductors; they are “in-between” materials. They have neither copper’s good conductivity nor plastic’s ability to insulate.
Semiconductors’ conductivity changes with the environment. For instance, energy, stress, or heat change semiconductor electrical conductivity.
When light falls on a semiconductor, electricity is generated. This is why we can only make solar cells from semiconductors.
Photoelectric Effect
Sunlight is a mixture of light of varying energy levels. High energy light excites the electrons in a semiconductor. This is known as the “photoelectric effect.”
Although electrons typically orbit an atom, excited electrons will break free of their atoms. But the excited electrons only move if the energy is high enough.
Imagine throwing money onto the ground at Times Square: if you only threw down a nickel, few people would be excited and pick it up. Same for electrons.
However, if you threw down $100 bills, people would run to grab them! Similarly, high-energy light excites electrons. This is the principle of the photoelectric effect.
Directing Electron Flow
Unfortunately, excited electrons just move around randomly. They have no overall direction, just like people running after scattered money.
However, if you threw $100 bills at one end of Times Square, people would run towards that side of the square.
Similarly, solar cells need to direct the electron flow towards a common goal. In solar cells, the electrons are forced to one side of the cell by an electrical force.
Solar cell designers pair together two different kinds of silicon to achieve this force. One kind (“p-type”) attracts electrons, and the other kind (“n-type”) repels them:
- P-Type silicon has fewer electrons and more “electron holes.” This attracts electrons.
- N-Type silicon has more electrons and fewer “electron holes.” This repels electrons.
Which Materials Direct Electron Flow?
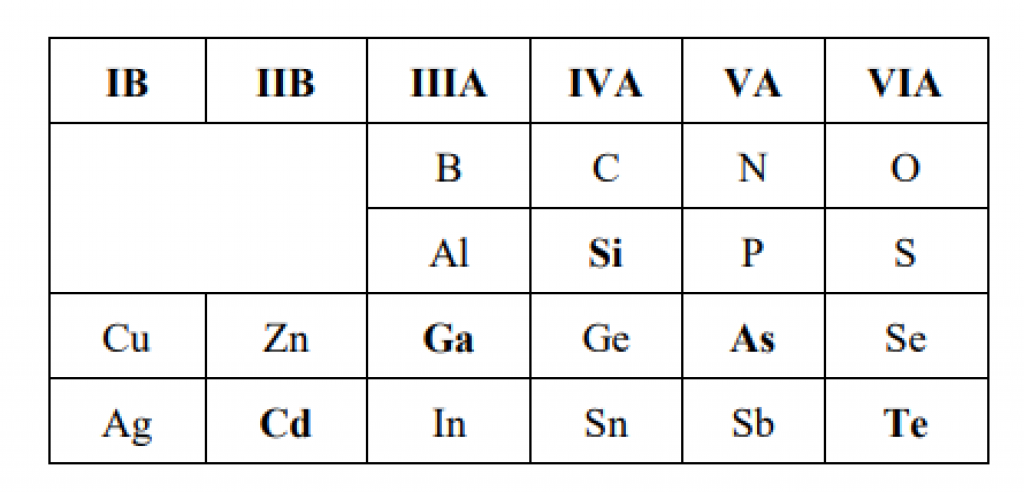
Source: Applied Photovoltaics, 3rd Edition, Wenham, et al.
The periodic table of the elements is a map of all known elements. The table arranges the elements into columns. Elements in the same column all have the same number of electrons in their outermost shell.
For example, Group 4 elements have four outer electrons; Group 3 elements have three outer electrons. And Group 5 elements have five outer electrons.
Since silicon (Symbol: Si) is a Group 4 element, it has 4 outer electrons. When boron (Symbol B, Group 3) is added to silicon, the boron atoms are missing one electron. This lack of electrons attracts electrons!
Similarly, adding phosphorus (Symbol P, Group 5) to silicon adds atoms that have an extra electron. This excess of electrons repels electrons!
This is how solar cell designers create P- and N-type silicon: they add in Group 3 and Group 5 elements.
This is also how solar engineers design thin-film cells that are not silicon-based. Designers will always pair elements from opposite groups.
For example, you may have spotted above that Cadmium Telluride is a thin-film solar cell. Designers pair cadmium (Group 2, 2 outer electrons) with Tellurium (Group 6, 6 electrons). The goal is to mix elements to mimic silicon’s four outer electrons.
Directing Electron Flow
Back to P-type and N-type silicon.
As the image below shows, P-type silicon attracts the electrons. N-type silicon repels the electrons.
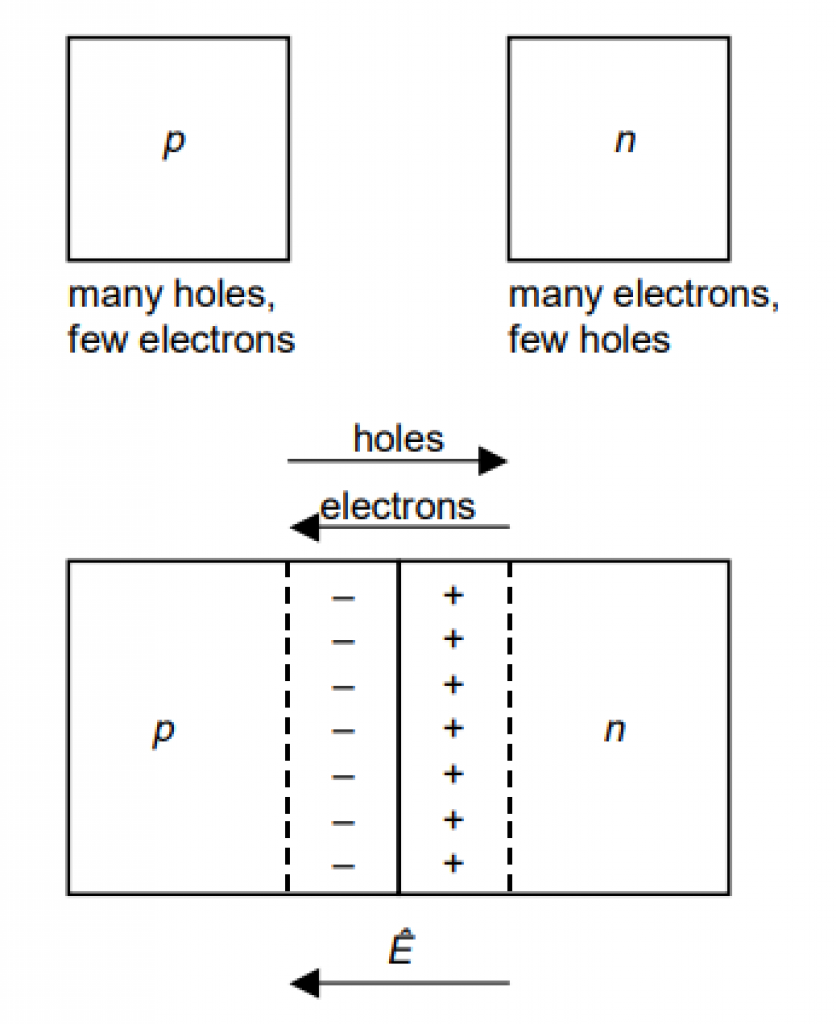
Source: Applied Photovoltaics, 3rd Edition, Wenham, et al.
Electricity is a flow of electrons. Therefore, the electrons keep flowing as long as light falls on the solar cell.
Cell Design

Silicon solar cells are made of N-type silicon and P-type silicon to direct electron flow. The top half of the cell is N-Type, and the bottom half is P-Type, like a Keebler wafer.
This directed flow of electrons from one half to another creates an electric field across the cell. Bare metal contacts embedded into the front and back of the solar cell then channel the flowing electrons.
Bringing It Together: Solar Panels
Surprisingly, individual solar cells produce a tiny amount of energy. Therefore, solar cells are wired to their neighbors to create a “daisy chain” of cells. The wiring groups the cells into electrical chains. This amplifies the electrical power of the cells.
These groups of cells are then sealed inside plastic laminate to keep out moisture and dirt. In addition, a transparent glass top sheet and a back sheet further protect the cells.
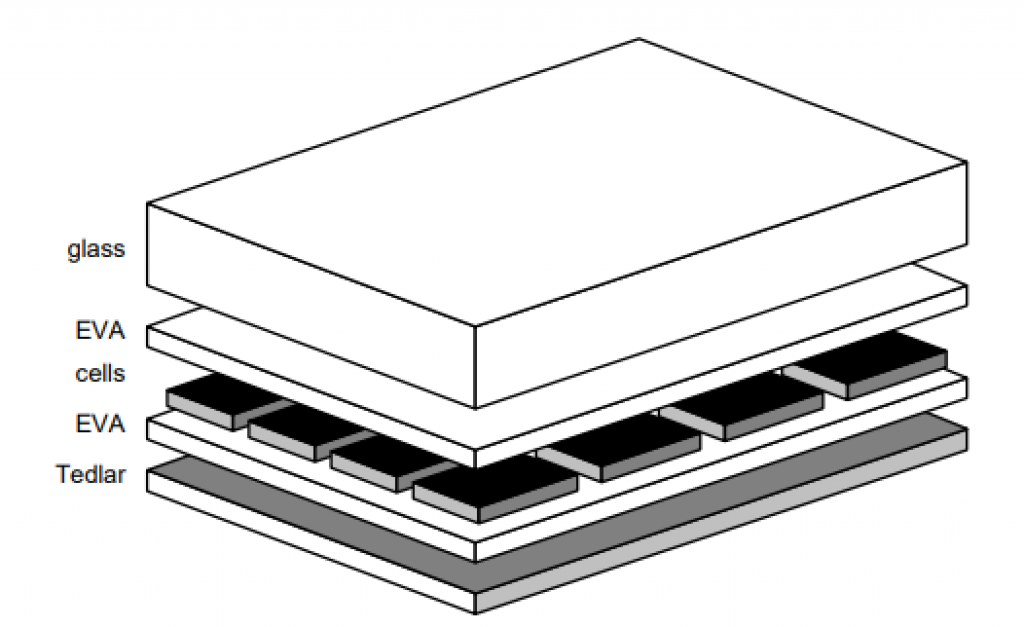
Source: Applied Photovoltaics, 3rd Edition, Wenham, et al.
Finally, a metal frame (to stiffen the panel) and a junction box with two cables complete the panel. The two cables — one positive, one negative — allows the panel’s electricity to be used.
Summing Up
In summary, thin-film cells are similar to crystalline silicon cells in their design and use of the photoelectric effect.
Both types of cells:
- Are made of semiconductors.
- Harness the photoelectric effect.
- Must direct electron flow so that electricity can flow
- Must be weather protected and mechanically tough
The big difference between silicon cells and thin-film cells is the type of semiconductor they’re made out of.
Now let’s cover the specifics of thin film solar cells.
What Are Thin-Film Solar Panels Made Of?
Like silicon solar cells, thin-film solar cells are made of a sandwich of semiconductors.
The core design of thin-film cells is also like silicon solar cells:
- A semiconductor layer that repels electrons (like the silicon N-type)
- A semiconductor layer that attracts electrons (like the silicon P -type)
- A means of channeling electron flow
Unlike silicon cells, thin-film cells do not have metal contacts on top and bottom. Instead, they have oxide or metal layers that have good conductivity. These oxide/metal layers are applied above and below the semiconductor layers.
There are three major types of thin-film cells:
- Copper Indium Gallium Selenide (known as “CIGS”)
- Cadmium Telluride (known as “CdTe”)
- Amorphous Silicon (known as “a-Si”)
The most critical semiconductor in the sandwich gives its name to each thin-film type.
CIGS
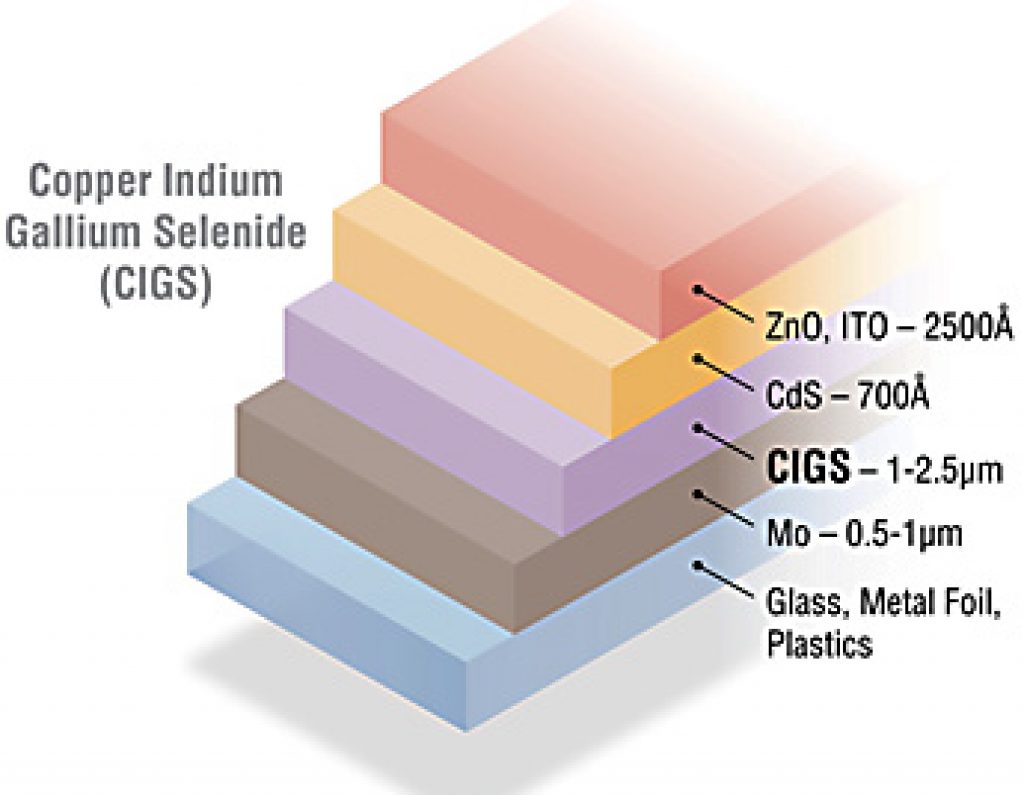
Source: NREL.gov
Copper Indium Gallium Selenide, or CIGS for short, is one of the most common thin-film solar cells.
From top to bottom, a CIGS cell is made of:
- A zinc oxide (ZnO) layer that is conductive
- A cadmium sulfide (CdS) layer which repels electrons
- A Copper Indium Gallium Selenide (CIGS) layer which attracts electrons
- A conductive molybdenum layer
Indium is a rare element and makes CIGS less desirable and less sustainable.
CdTe
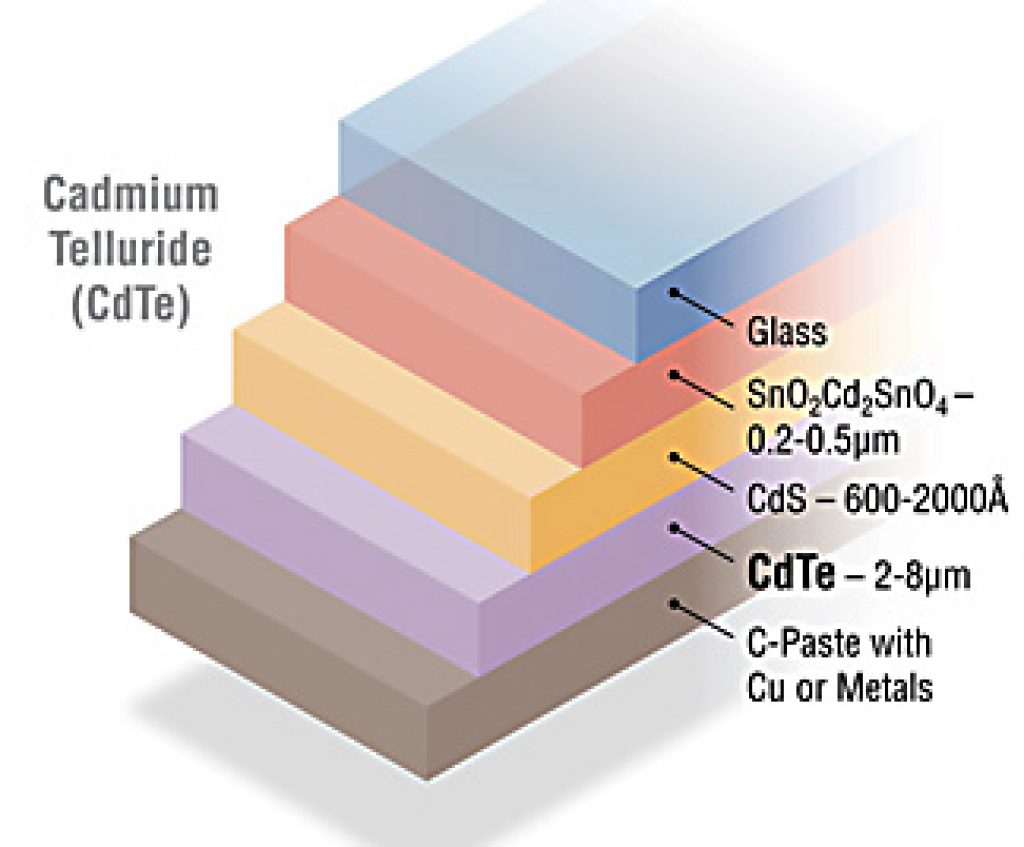
Source: NREL.gov
Cadmium telluride (CdTe) is the most widely used thin-film technology, with a 50% market share amongst thin-film cells.
From top to bottom, a CdTe cell is made of:
- A tin oxide (SnO2) or cadmium stannate (Cd2SnO4) layer that is conductive
- A cadmium sulfide (CdS) layer which repels electrons
- A cadmium telluride (CdTe) layer which attracts electrons
- A layer of carbon paste infused with copper that is conductive
Cadmium is a toxic heavy metal, like mercury. It can accumulate in food chains, and for this reason, researchers are looking for non-toxic substitutes.
a-Si
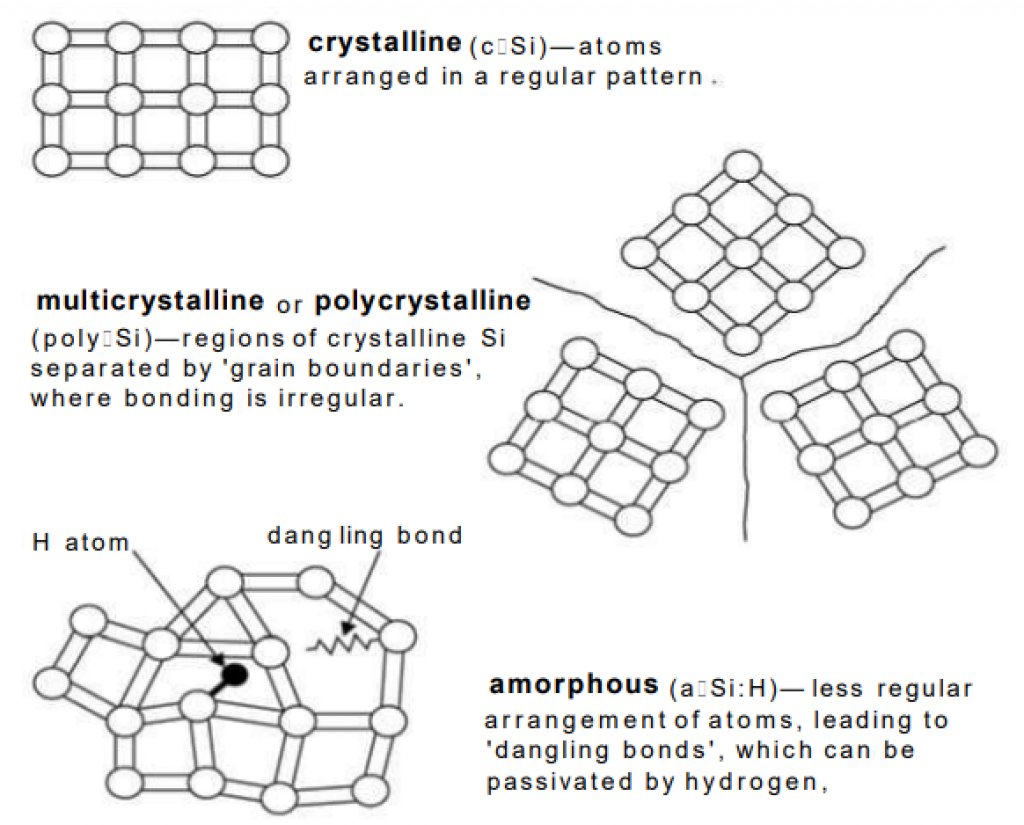
Amorphous Silicon (“a-Si”) is the third major kind of thin-film solar cell. In contrast to crystalline silicon solar cells, a-Si does not have an overall crystal structure (see chart above).
Low efficiency is a major challenge for a-Si solar cells. However, the cells are made of silicon, a non-toxic material.
How Do Thin-Film Solar Cells Compare to Silicon Cells?
Thin-Film Vs. Silicon: Efficiency Comparison
Like any machine, solar cells are more or less efficient at their job. Solar cell efficiency is the success rate of converting sunlight (in Watts) into electricity (also in Watts).
So a 12% efficient cell is more efficient than a 5% cell. Given the same light, a higher efficiency cell generates more energy over time.
Thin-film solar cells are typically less efficient than silicon solar cells, but it depends on the specific cell.
The table below gives you an idea of the relative differences, but solar cell efficiency has also improved over time. Research has yielded higher efficiency thin-film cells.
| Solar Cell Material | Solar Cell Efficiency |
|---|---|
| Mono-crystalline silicon | 12-20% |
| Poly-crystalline silicon | 11-20% |
| Copper Indium Gallium Selenide (CIGS) | 10-13% |
| Cadmium Telluride (CdTe) | 9-18% |
| Amorphous Silicon (a-Si) | 5-7% |
Although the explanation is highly technical, all solar cells lose some efficiency as the cell temperature increases. Thin-film solar cells maintain their efficiency better than silicon solar cells.
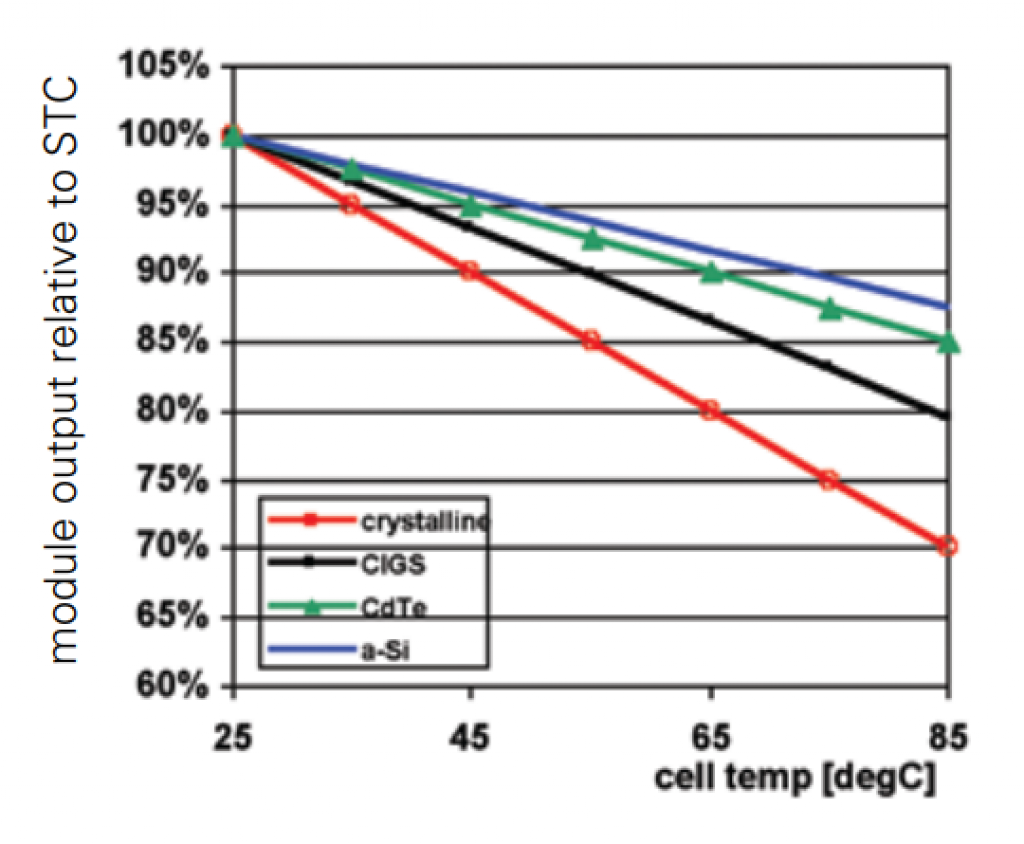
Source: Singapore Energy Market Authority
Examining the chart above from left to right:
- Crystalline solar (red line) relative efficiency drops from 100% to 70%
- The three thin-film cells (black, green, and blue) relative efficiencies drop from 100% to 80-87%
Since solar panels last a long time, this 10% difference makes a big difference over the long run.
Because thin-film solar cells maintain efficiency as the cell temperature rises, they are better suited to hot locations.
Thin-Film Vs. Silicon: Cost Comparison
Thin-film cells are cheaper than silicon cells. This is because thin-film cells need less material per square foot of panel.
To illustrate, the table below shows the indicative price difference for thin-film and silicon solar cell panels.
| Solar Cell Material | Indicative Panel Cost Per Watt |
|---|---|
| Mono-crystalline silicon | $1.50 |
| Poly-crystalline silicon | $1.50 |
| Copper Indium Gallium Selenide (CIGS) | $0.60 |
| Cadmium Telluride (CdTe) | $0.40 |
| Amorphous Silicon (a-Si) | $0.70 |
The cost advantage of thin-film cells has resulted in niche uses, although silicon solar cells hold a 90% market share.
What Are Thin-Film Solar Panels Used For?
Application 1: Utility Scale Plants In Hot Places
As we covered earlier, thin-film cells lose less performance in hot climates. Therefore, thin-film solar panels will perform better in hot locations.
Another advantage of thin-film solar panels is that their light weight makes them better suited to tracking solar arrays. Lighter panels allow the tracking structure to be lighter and cheaper.
Thin-film cells are generally lower in their efficiency than silicon cells. Therefore, thin-film cells need more area for given power output.
This is not a problem for utility-scale solar plants because project developers usually build utility-scale plants on cheap land.
In summary: hot, sunny places with cheap land are ideal for thin-film solar panels, such as the deserts of the Southwest.
Application 2: Flexible, Cheap, Or Light Products

Thin-film solar panels are mechanically flexible. For example, curved building facades or solar carports with curved roofs can use thin-film solar panels. Even buses have thin-film solar panels embedded in their roofs!
Thin-film cells are suitable for many consumer products, being light and cheap — for example, watches or calculators with integrated solar cells.
Even peel-off stickers can include thin-film cells.
Are Thin-Film Solar Panels Flexible?
Thin-film solar panels are flexible because the semiconductors on thin-film cells are extremely thin; they can bend to a high degree without breaking.
How Long Do Thin-Film Solar Panels Last?
In short, thin-film solar panel manufacturers offer warranties similar to c-Si solar panel manufacturers.
This suggests that thin-film solar panels last just as long as other cell types. Thin-film manufacturers claim their products have a lower long-term degradation than silicon cell panels.
However, the solar industry generally regards thin-film solar panels as having a shorter lifespan than crystalline solar panels.
With this in mind, you should check thin-film solar panel durability by reviewing panel datasheets. Contact your local suppliers and ask for the datasheets for their products.
Each datasheet will list the warranty terms and the long-term predicted reduction in power output.
Final Thoughts
In summary, thin-film solar cells are cheap and flexible. They are part of the broader family of photovoltaic solar cells and have a long track record.
In light of the advantages and disadvantages of thin-film solar, it may be the right choice for your project or product.

