Lithium titanate batteries have become an increasingly popular rechargeable battery, offering numerous advantages over other lithium technologies.
Nowadays, you’ll find them in various applications, from electric vehicles (EVs) to consumer electronics.
With high charge/discharge rates, considerably long cycle life, low internal resistance, wide working temperature, and increased safety, this battery’s popularity will only grow in the near future.
In this article, we provide an overview of lithium titanate batteries and explain their key features, applications, and benefits. Additionally, we discuss the potential drawbacks of using this type of battery.
Table of Contents
What are lithium titanate batteries?
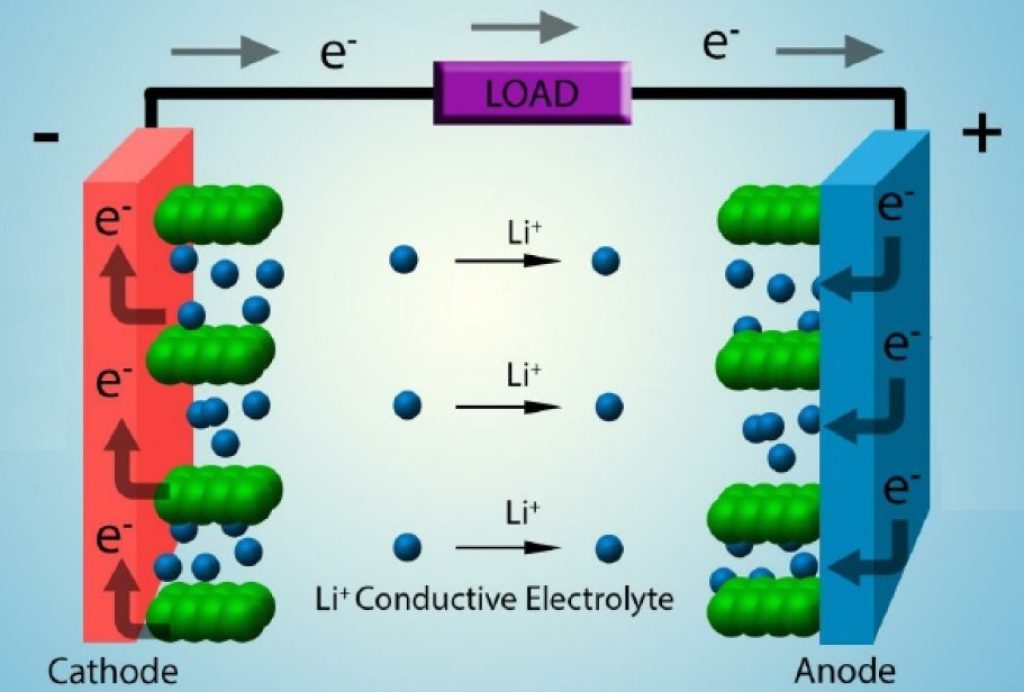
Lithium titanate, or lithium titanate oxide (LTO) batteries, are rechargeable batteries that use lithium titanate oxide as the anode material.
These batteries fall under the lithium titanate classification. Their chemistry is based on the exchange of lithium ions between the cathode and the anode.
Source: Adapted from “3-D Micro and Nano Technologies for Improvements in Electrochemical Power Devices.”
However, there’s a critical difference between lithium titanate and other lithium-ion batteries: the anode.
Unlike other lithium-ion batteries — LFP, NMC, LCO, LMO, and NCA batteries — LTO batteries don’t utilize graphite as the anode.
Instead, their anode is made of lithium titanate oxide nanocrystals.
This unique feature significantly impacts this battery’s properties.
But why? What’s so special about this anode?
How Does The Anode Material Impact The Battery’s Properties
In a lithium-ion battery, ions move from one electrode to another.
The direction in which these ions move depends on whether you’re charging or discharging the battery. During charging, the lithium ions move from the cathode to the anode.
Lithium ions can enter and exit the anode’s structure. The speed/rate at which this happens depends on the anode’s ability to “accommodate” these lithium-ions. In chemistry, the term for this “accommodation” is intercalation.
Lithium titanate as the anode material
Intercalation
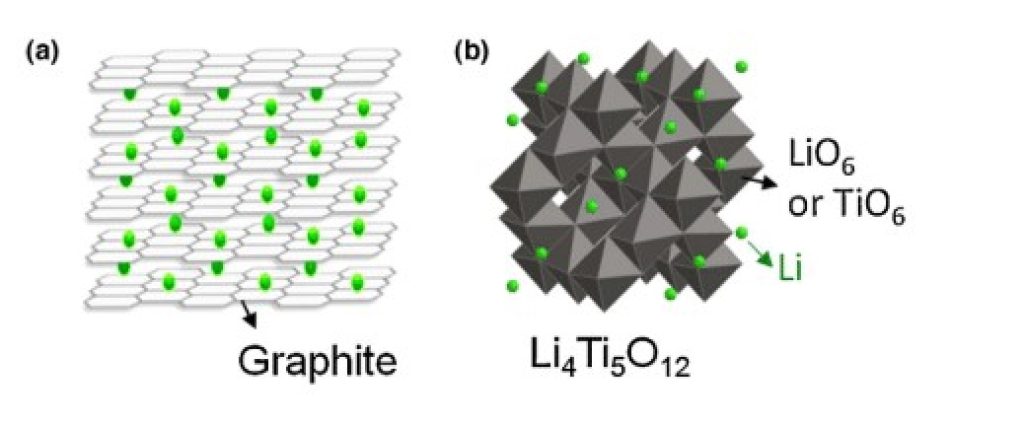
Graphite is the prime anode material for most lithium-ion batteries. This is due to its low cost, availability, and convenient electrochemical properties.
However, its lithium intercalation capacity is relatively poor.
Now, guess which material is great for lithium intercalation? That’s right, lithium titanate oxide.
Source: Adapted from “Li-ion Batteries for Electric Mobility“
Another problem with graphite as the anode material is its volume changes during charge/discharge cycles. Over time, this expansion/contraction irreversibly damages the battery cell’s structure, limiting its lifespan.
Conversely, lithium ions going in and out of the anode’s structure during charge and discharge has almost no effect on lithium titanate structures.
Therefore, the LTO anode practically doesn’t suffer any volume changes during cycles, minimizing possible structure degradation, improving battery performance, and ultimately extending battery life.
Low degradation rate
The LTO anode’s structure facilitates lithium ions entering and exiting, allowing electrons to enter and exit the anode faster.
This makes fast charging/discharging (higher current) much safer for LTO batteries than graphite as the anode since lithium dendrites are less likely to form, avoiding degradation and possible short-circuit.
Moreover, the anode’s properties minimize the risk of SEI film formation and lithium plating — this helps avoid capacity loss.
Finally, LTO batteries charge well under low temperatures and maintain thermal stability under high temperatures.
Is lithium titanate good for solar applications?
The answer here depends on what you’re looking for in a solar battery.
LTO batteries can provide a high charge/discharge rate. This makes them suitable for applications that require fast charging and a high current burst.
However, their energy density (energy stored per volume) is relatively low, so you’d need an extensive system to achieve a high capacity.
Therefore, if you have limited/space for your solar battery bank, you’d be better off choosing battery storage with higher energy density, such as lithium iron phosphate (LiFePO4) batteries.
That said, if your energy demand is low, an LTO battery would be worthwhile, as it requires fewer solar hours to charge.
Another advantage of LTO batteries is their high resistance to extreme temperatures. For instance, an LTO battery’s performance is resistant to being left in a warm place/under sunlight — though it’s best to avoid doing this.
Ultimately, lithium titanate batteries make worthwhile solar batteries if you’re priorities are:
- Cycle life.
- Charge/discharge times.
- Safety.
However, if you desire a large capacity and don’t care much about high charge/discharge rates, an LTO battery won’t be the best solar battery technology for your needs.
Battery comparison: LTO vs. LFP
| Properties | LTO (Lithium Titanate Oxide) | LFP (Lithium Titanate Oxide) |
|---|---|---|
| Charge/Discharge Rate | Up to 10 C | Limited to 1C |
| Cycle Life | 5.500 to 7.300 cycles | 2.000 to 4.000 cycles |
| Nominal Voltage | 2.4 V | 3.3 V |
| Energy Density | 177 Wh/L | 325 Wh/L |
| Specific Energy | 60–110 Wh/kg | 90–160 Wh/kg |
| Working Temperature Range | -40°C to 50°C | 5ºC to 40ºC |
Limitations of LTO batteries
One of the primary limitations of lithium titanate (LTO) batteries is their cost. They are more expensive than other lithium-ion batteries, such as lithium iron phosphate.
Another limitation is their capacity. LTO batteries have a lower energy density than other types of batteries, so they might not be the best option for energy storage where space is limited.
Finally, LTO batteries are not as widely available as other types of batteries, making them harder to source.
Are lithium titanate batteries safe to use?
Lithium titanate batteries are considered the safest among lithium batteries.
Due to its high safety level, LTO technology is a promising anode material for large-scale systems, such as electric vehicle (EV) batteries.
They are non-flammable, non-explosive, and do not release toxic gases when overcharged or heated, reducing the risk of fire and explosion.
Overall, LTO batteries are a safe and reliable choice for many applications.
Here’s an interesting video made by Yinlong to show the safety of their LTO batteries:
What is the lifespan of lithium titanate batteries?
Discussing battery lifespan is not a simple task — it depends on many variables and can vary greatly depending on usage habits.
Typically, a battery reaches its end of life when its capacity falls to 80% of its initial capacity.
That said, lithium titanate batteries’ capacity loss rate is lower than for other lithium batteries. Therefore, it has a longer lifespan, ranging from 15 to 20 years.
These numbers translate to around 5,500 to 7,300 cycles, considering one cycle per day.
Related Article: Lithium Battery Cycle Life
Do lithium titanate batteries need a BMS?
Using a battery management system (BMS) in LTO batteries is highly recommended.
Even though LTO batteries are more resistant than other lithium batteries — especially when operating at high temperatures — you should still use a BMS.
A BMS monitors and manages a battery’s properties, such as voltage, current, and internal temperature, to ensure optimal charging and discharging processes. Additionally, it protects a battery’s structure.
In short, a BMS helps maximize a battery’s lifespan and performance.
Therefore, it’s essential for any battery system, including LTO batteries.
Final thoughts
Lithium titanate batteries offer many advantages over other lithium-ion chemistries, including:
- Longer cycle life.
- Increased safety.
- Wider working temperature range.
- Faster charge/discharge rates.
However, energy density is relatively low among these batteries. In addition, high C-rates inevitably impact the battery’s capacity over time.
Ultimately, it’s essential to consider the advantages and disadvantages of LTO batteries before deciding which type of battery is best for your application.
Currently, Toshiba and Yinlong are the leading LTO battery providers. But there are many other companies manufacturing and investing in research and development of LTO batteries, primarily due to their promising applications in the field of EVs.
Should you have questions regarding this topic, please reach out to us in the comments section below, or follow us on Twitter.

