If you don’t want to find yourself with a “dead battery suddenly,” then learning about battery capacity is an absolute must!
Since most electronic devices are powered by batteries nowadays, it’s only logical that we ask ourselves a few questions about these energy storage technologies, such as:
- How do they work?
- What can they power?
- How long do they last?
- What can I do to make them last longer?
If you start to look for answers to these questions, you’ll notice that terms like “battery capacity,” “amps,” “Ah,” and “kWh” are tossed around left and right.
Therefore, before you explore the complex yet exciting world of batteries, you should start with the basics. And the battery capacity is right at the top of the list.
In this article, we prepared a comprehensive guide to understanding battery capacity, once and for all.
Table of Contents
What Is Battery Capacity?
Battery capacity is defined as the total amount of electricity generated by electrochemical reactions in the battery.
It’s directly proportional to (and limited by) the usable amount of material inside the cell that actively participates in the redox reactions, providing charge (electrons).
Therefore, if some of this material is somehow consumed by irreversible reactions within the battery, it can no longer be used to convert chemical energy into electrical energy. Thus, the battery’s capacity decreases.
Conversely, the more electrode material in the cell, the greater its capacity.
A battery’s capacity is usually rated in Amp-hours (or mAh, for small-scale batteries). It expresses the total amount of current a battery can deliver in 1h until its voltage drops to a specific value for each cell (cut-off voltage).
Battery Capacity Rating Standards Are Misleading
In reality, the Ah rating of your battery is what your battery can deliver in ideal conditions. This value is lower than what manufacturers advertise in any other condition.
For example, a battery rated 200Ah can deliver 200A for 1h, in ideal conditions.
Likewise, it can theoretically deliver 100A for 2h, 50A for 4h, 10A for 20h, 2A for 100h, etc.
In reality, though, these numbers aren’t accurate. Several factors decrease the rated battery capacity, such as temperature, rate of discharge, and Peukert’s Law.
And of course, that doesn’t mean you can draw 12,000 Ah in 1 minute from a 200Ah battery because the redox reactions rate (the speed at which redox reactions take place) limits the conversion of chemical energy to electrical energy.
In addition, for lead-acid batteries, the Ah rating is usually given at a “20-hour rate”. So a 100Ah battery can only provide this 100Ah capacity during a 20h period (5A for 20h). If you connect the same battery to a 100A load, it might only last for a few minutes. Anything faster than that, and the capacity of this battery is no longer 100Ah; it’s actually much lower.
The higher the rate of discharge, the higher the internal resistance inside the cells, and the more energy wasted via heat. Conversely, the lower the drain, the more efficient the battery is.
In conclusion, the way you discharge your battery (at a high current, for example) impacts the battery’s actual capacity.
How Do You Calculate Battery Capacity?
Experimentally, battery capacity can be determined by measuring the discharge duration when the battery is discharged at a constant current.
Knowing the discharge current and the discharge duration, you can calculate battery capacity (in Ah) as follows:
Charge Capacity (Ah) = Current (A) x time (h)
This way, “Amp-hours” (or Ah) refers to the charge capacity of your battery. It expresses the amount of current that the battery can deliver in 1h, until its voltage drops to a point where it can no longer “push” enough electrons (produce current).
Now, here’s something to get you thinking: the image below shows a Li-ion battery pack of 3 single cells (on the left) and a Li-ion single cell (on the right).

Both batteries have a rating of 3500mAh. Does that mean they both provide the same amount of energy, even though one is 3 times larger than the other?
You guessed right — of course not!
That’s why another way of expressing battery capacity (and this is a more accurate way) is in terms of energy capacity (Wh). It represents how much energy can be stored in/provided by a battery in 1 hour.
Energy capacity (Wh) = Voltage (V) x Amp-hours (Ah)
Now that we have this information, let’s go back to the above image of the two Li-ion batteries. The left one is rated 3500mAh and 11.1V, so the energy it can store is given by:
Energy = 11.1V x 3500mAh = 38,850mWh = 38.85Wh
On the other hand, the single Li-ion cell on the right has a rating of 3500mAh and 3.7V, so the energy it can store is:
Energy = 3.7V x 3500mAh = 12,950mWh = 12.95Wh
This shows that, even though both batteries have the same charge capacity (in mAh), they have different energy capacities (in Wh). As expected, the larger battery (on the left) has a higher energy capacity than the single cell to deliver more energy at the same charge capacity of 3500mAh.
Usually, appliances are rated based on how much power they require to function (Power (W) = voltage x current).
Therefore, estimating the battery capacity in terms of energy capacity (Wh) can make things much easier to calculate how much battery capacity you need to power your appliances.
How Much Battery Capacity Do You Need?
It depends on your energy demand. Appliances are rated with a certain voltage and current. Using an amount higher than the recommended for these ratings will damage your appliance.
You can calculate how much battery capacity you need to meet your energy demands by checking the power rating (in Watts) of every appliance you wish to power with a certain battery.
Once you know all the power ratings of the appliances you wish to power, you need to take into account the amount of time (in hours) each appliance must be powered.
For example, you need to power a fridge 24h a day. Meanwhile, you only need to power a coffee maker for a few minutes a day.
Multiplying the power ratings (in Watts) by the amount of time you need to power your appliances, will give you the total amount of energy you’ll need (in watt-hours).
With this number in your hands, you just have to find a battery with specifications compatible with your needs.
For example, let’s take a 24V 200Ah LiFePO4 battery. You can calculate its energy capacity by multiplying its voltage(V) by its nominal capacity (Ah), so 24V x 200Ah = 4.8 kWh.
Because it isn’t recommended to fully discharge your battery, you should only use about 80% of this capacity. So, 80% of 4.8 kWh equals 3.84 kWh.
So with this battery, you can meet the total energy demand of up to 3.84 kWh.
How To Measure Battery Capacity With A Multimeter?
You can use a multimeter to measure (indirectly) battery capacity.
More precisely, you can use a multimeter – or a voltmeter – to measure your battery’s voltage. Then, using the correct chart, you can use the voltage you just measured to determine your battery’s current capacity.
You might be wondering, “How is this possible?”
Here’s the answer:
As previously mentioned, during discharge, the battery’s voltage decreases. Depending on the battery’s chemistry (and the rate at which you discharge your battery, called “C-rate”), you can consider this decrease linearly proportional to the state of charge (thus, the capacity left).
As an example, take a look at the graph below. It shows the discharge curve of a 12V LiFePO4 (Battery Voltage (in V) x State of Charge (in %)):
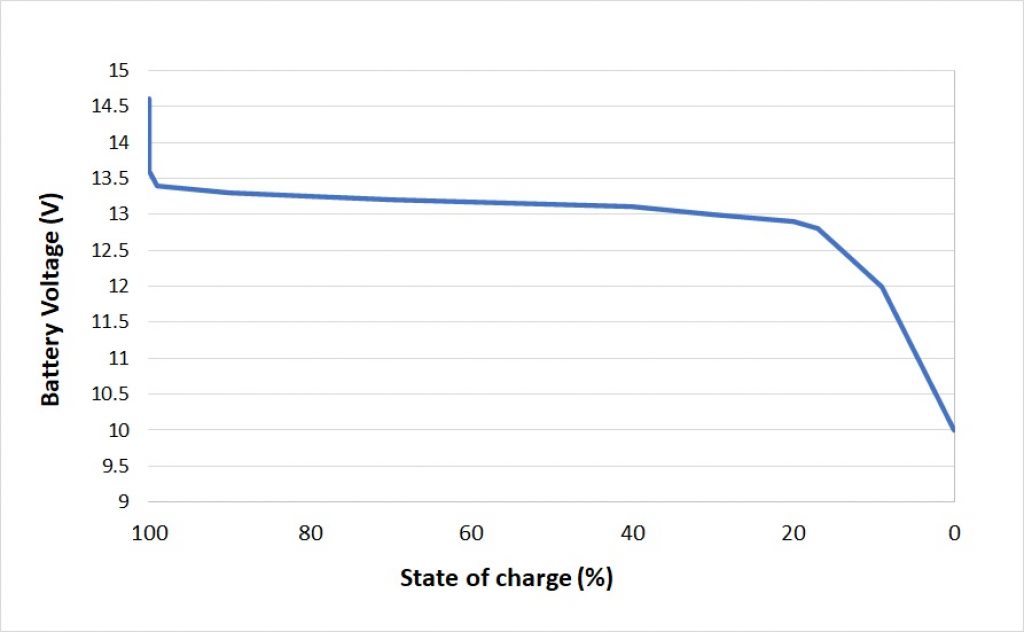
Source: climatebiz
As you can see, the voltage decreases as the state of charge decreases. Not only that, you’ll notice that the shape of the curve is almost linear until about a 20% state of charge (discharging a lithium battery below 20% is not recommended anyway).
The fact that this variation is almost linear means that the voltage and the state of charge of a battery decrease proportionally. You can use this information to determine a battery’s state of charge (and therefore its capacity) by measuring its voltage.
In the following table, you can see the values of Battery Voltage and State of charge used to plot the graph above:
| VOLTAGE | STATE OF CHARGE |
|---|---|
| 14.6V | 100% (charging) |
| 13.6V | 100% |
| 13.4V | 99% |
| 13.3V | 90% |
| 13.2V | 70% |
| 13.1V | 40% |
| 13.0V | 30% |
| 12.9V | 20% |
| 12.8v | 17% |
| 12.5V | 14% |
| 12.0V | 9% |
| 10.0V | 0% |
Consult your battery’s data sheet and check its discharge curve.
So how exactly can you do this?
Here’s The Step-By-Step Process:
- Get a multimeter and set it up to measure the voltage of a direct current supply (remember: batteries provide direct current, DC). It is represented by a V with a ⎓ symbol on top (as opposed to a ~, for AC)
- Also, select a reading voltage higher than the nominal voltage of your battery. So, for a 12V battery, choose 15V or 20V (the scale varies for different multimeters)
- Now, connect the red probe to the battery’s positive terminal and the black probe to the battery’s negative terminal.
- Look for your battery’s voltage x capacity (or state of charge) chart (it differs depending on the battery’s chemistry). You can usually find it in your battery’s datasheet.
Example
Here’s the datasheet of a RELiON 12V 200Ah LiFePO4 battery:
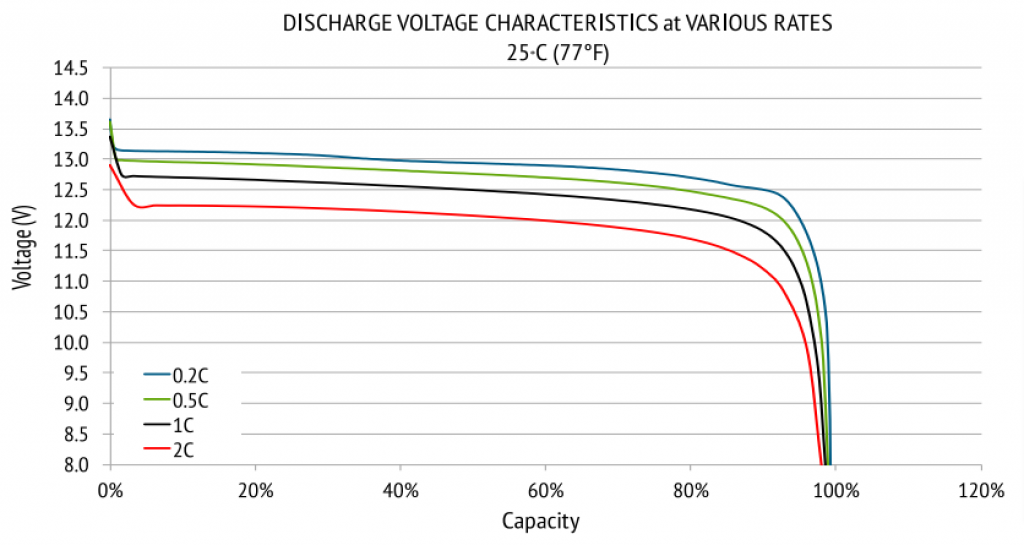
Source: relionbattery.com
In conclusion, you can use a multimeter to measure your battery’s voltage and then use this information to determine your battery’s capacity, using a voltage x capacity chart (according to your battery’s chemistry and specifications).
Additionally, you can use multimeters to measure current, up to a point. On most multimeters, the highest current you can measure is 10A. You can’t feed very high currents through a multimeter because their usual 4 mm sockets and wires cannot carry high currents.
However, if you want to measure the battery capacity (in Ah) of a small battery – a 1,2V 1600mAh Ni-MH battery, for example -, you can use this feature of your multimeter.
In this case, you have to:
- Set up your multimeter to measure current (expressed by A)
- Connect the battery to a load (a resistor, for example)
- Connect the probes to your battery’s terminals
- Start a stopwatch to measure elapsed time
- Check the current reading about every hour
- When the current is about 70% of the original reading, stop the stopwatch
- Calculate the battery’s mAh rating by multiplying the time elapsed (in h) by the multimeter’s initial current reading (in A).
For example, if the initial current was 160mA and 10h elapsed, the Ah rating would be 160mA x 10 h = 1600mAh.
This way of measuring isn’t practical, though, since it wastes the battery’s capacity during the measurement.
For more information, this article can be pretty helpful.
Battery Capacity mAh (Explained)

Source: fenixlighting.com
We already covered that battery capacity is usually defined in terms of charge capacity, which is how much current a battery can provide in 1h, until it can no longer provide current.
We also saw that the unit for charge capacity is Amp-hours (Ah).
Now, you might notice that for smaller batteries — such as rechargeable AA batteries — the capacity is provided in mAh.
The “m” in mAh is simply a metric prefix.
A metric prefix is a unit prefix that precedes a basic unit of measure to indicate a multiple or submultiple of the unit. The prefix “m” stands for milli, from the Latin mille, meaning one thousand.
With that said, mAh is one-thousandth (10-3) of Ah.
So next time you see a battery rated 3500mAh, you can easily convert it to Ah, by multiplying 3500mAh x 10-3 = 3.5 Ah. Another way of converting it is to divide the 3500mAh by 1000, giving you 3.5 Ah.
Battery Capacity Wh (Explained)
Sometimes, you see battery capacity being described in terms of energy capacity. This amount of energy is measured in Wh.
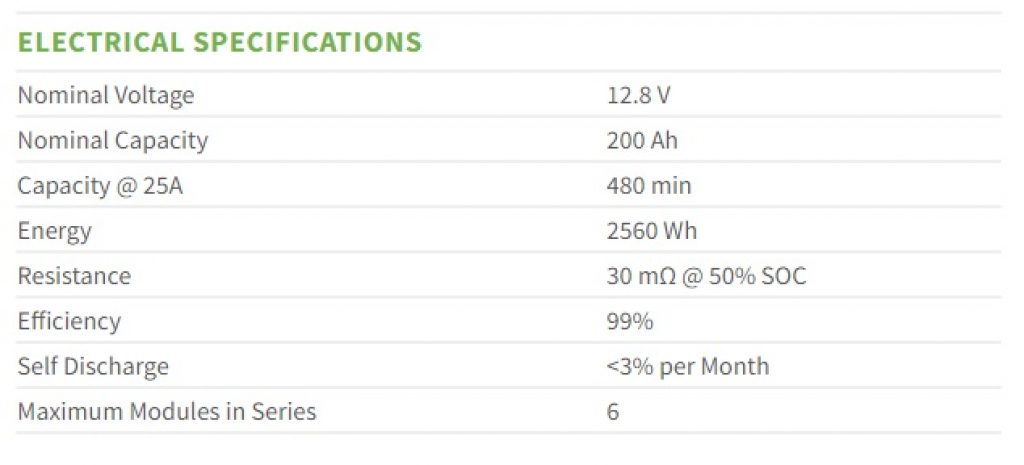
Source: relionbattery.com
You’ll notice in the above image that the rated energy of this battery is 2560Wh.
Here’s how you calculate this energy:
In physics, you can calculate energy using Energy = Power x time. In turn, Power = voltage x current. So, replacing the expression for power in the energy formula, we get: Energy = voltage x current x time
Let’s Analyze These Units
- The unit of energy is Joules (J)
- The unit of voltage is Volts (V)
- The unit of current is Amperes (A)
- The unit of power in Watts (W)
- And, for this case, the unit of time in hours (h)
So, if we rewrite “Energy = voltage x current x time” in terms of its units, we’ll realize we can express energy (J) in terms of Watt-hours (Wh):
[J] = [V]x[A]x[h] -> [J] = [W]x[h]
In conclusion, you can also express battery capacity in terms of energy capacity, which is how much power a battery can store (thus, provide) in 1 hour.
This is a better way of understanding battery capacity because it takes the voltage into account (which varies during discharge). It is easier to compare with the power ratings of appliances, usually given in Watts (W).
Battery Capacity Ah (Explained)
You’ll often notice Amp-hours as the unit of battery capacity. It expresses the amount of current you can draw from a battery in 1h, until the battery’s voltage drops to a point where it can no longer provide enough current.
You can estimate how long your battery will provide current to your load if you know the current drawn by this load.
For example, if you have a battery rated 20Ah, and you connect it to a load that draws 2A, you’ll be able to provide current to this load for 10h.
You can use the expression below to calculate one of these parameters (Ah, A, and h), if you already know the other two:
Charge capacity (Ah) = current the battery provides (A) x the amount of time in which this current was provided (h)
Battery Capacity kWh (Explained)
As previously explained, Wh expresses the energy capacity of a battery. In other words, it expresses how much power the battery can provide in 1 hour, until it is drained.
Now, you’ll often find kWh to express this energy capacity. The “k” is also a metric prefix. It stands for kilo, and denotes one thousand (103 = 1000).
So, with that in mind, is it now easy to understand kWh. For example, if a battery is rated 2.4kWh, it means that its energy capacity is 2,4 x 103 Wh = 2400Wh.
Final Thoughts
With the fast advancements in technology, batteries rapidly became a basic need for all of us. It powers phones, laptops, cameras, vehicles, boats, medical equipment, and all sorts of electronic gadgets.
Because we are so dependent on these devices, understanding how batteries work and what they are capable of should probably be more common than it is.

